Roberta Tevlin, OAPT Editor, Teacher Danforth CTI
Roberta.tevlin@tdsb.on.ca
Edited by Margaret Scora
Lasers are quantum light sources and they are everywhere. But what is quantum about them? The PhET simulation is a great tool to give students a feel for the quantum process called stimulated emission.
What is so special about laser light?
The word ‘laser’ is an acronym for Light Amplification by Stimulated Emission of Radiation. It sounds dangerous, doesn’t it? It needn’t be. The last letter is not talking about nuclear radiation, it just refers to electromagnetic radiation and the laser could have been called a ‘lasel’ except that it doesn’t sound as good.
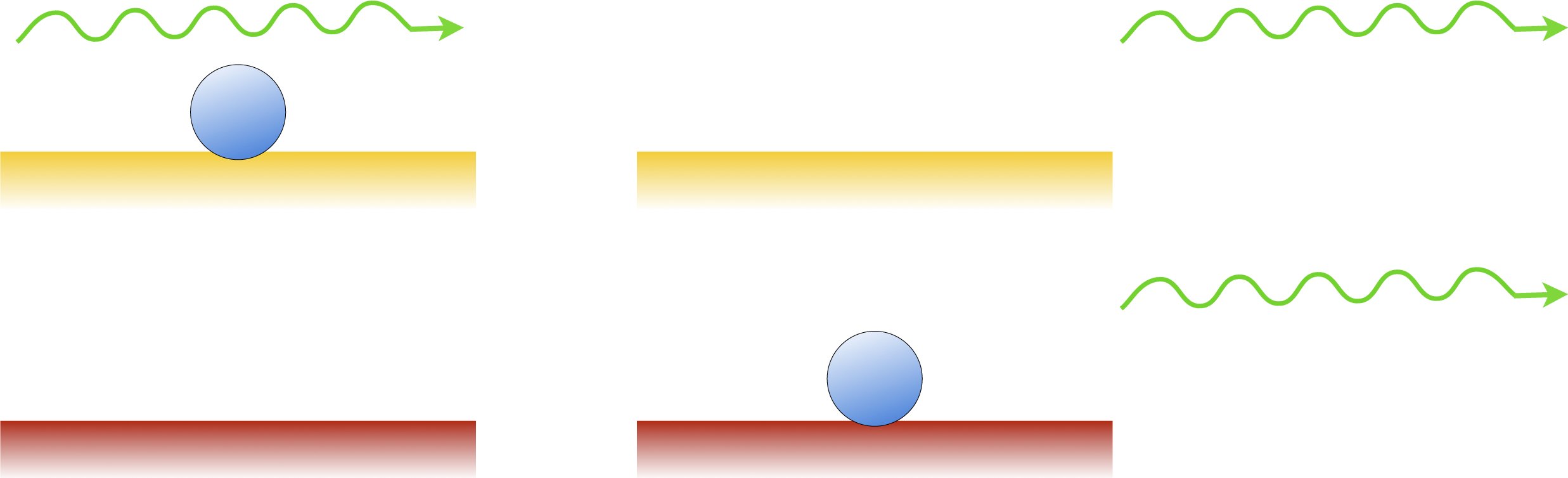
Consider a flashlight and a laser pointer. How do the photons of each device differ? Contrary to the common misconception, they are not necessarily more energetic, powerful or focussed. They are just more organized. In an ideal laser, all of the photons have the exactly the same frequency, direction, phase and polarization. This organization is a result of stimulated emission, a process that was predicted by Einstein. Normally, when an electron is in an excited state, it will spontaneously return to the ground state and emit a photon in a random direction. However, if a second photon with the same frequency comes by, it will stimulate the atom to emit its photon to match the original one. The difference is shown in the diagrams below.
Spontaneous emission
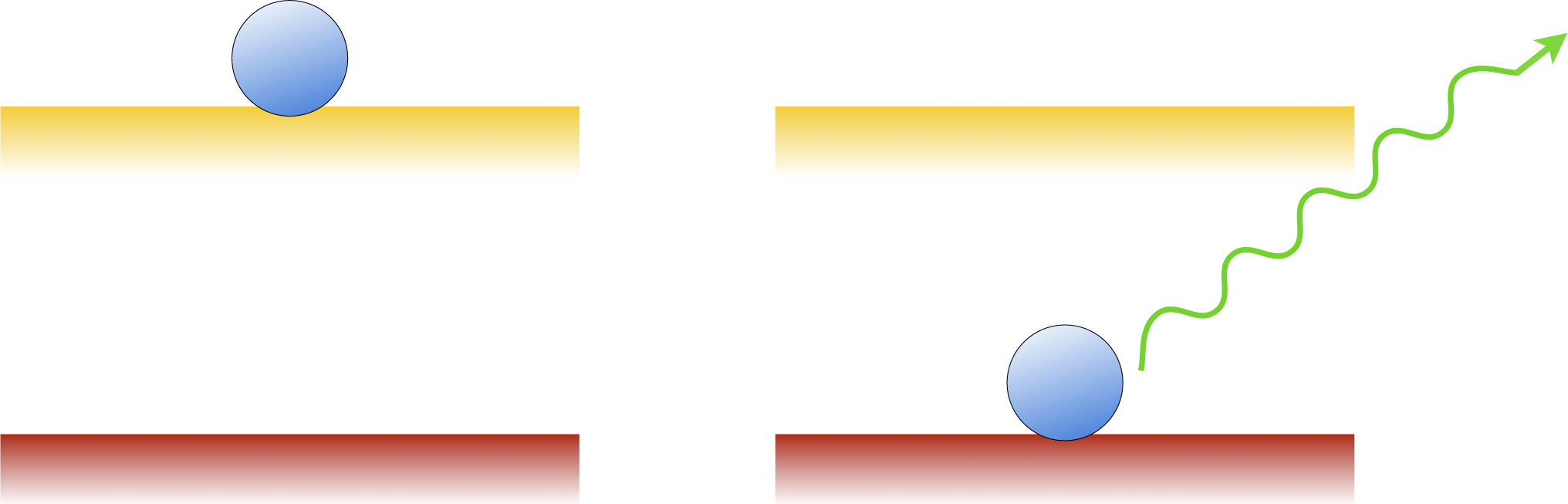 Stimulated emission
Stimulated emission
 Making Stimulation Emission easier to understand with Concrete Models
Making Stimulation Emission easier to understand with Concrete Models
Stimulated emission is a quantum process, but it would be helpful if we could present students with a concrete example of a macroscopic process that is similar to it. There isn’t one model that does it properly, but students can gain a deeper understanding of stimulated emission by examining two models that follow and discussing where they match and where they don’t.
Mechanical resonance has some similarities. You can demonstrate the resonance with matched tuning forks or by playing a trumpet near a guitar or piano. You can also have them watch this video that I found recently, of
32 Metronomes Demonstration of Resonance. Resonance does a good job of demonstrating the importance of matching frequencies but it does not show amplification.
An analogy that does show amplification is a box full of mouse traps that are all set to go off. If one of them is sprung, it will jump and set the others off, amplifying its usual effect. If you don’t have the patience to set this up yourself — and I certainly don’t! — you can watch the video
Mousetrap Fission. This demo shows amplification, but there is no matching of frequencies.
How do you make a laser?
Einstein described stimulated emission in 1917, but it wasn’t until 1957 that Charles Townes and Arthur Schawlow were able to harness this process. It was called a maser, because it produced microwaves — not visible light. They got the Nobel Prize for this work in 1964. The
PhET simulation is a fantastic tool to understand how a laser works.
When you open the simulation, you will see absorption and emission of photons by one atom. If you change the colour of the photons, the process will stop because the photons will no longer have the energy to match the jump from the ground state to the next level. If you increase the intensity, you will see more photons and you will also start to see photons moving to the right in pairs, showing that there has been stimulated emission. If you turn the lifetime of the excited state down, this stimulated emission stops, because the atom decays before the next photon form the lamp arrives. We now know how to get stimulated emission, but not a laser beam. This requires multiple atoms.
Go to the Multiple Atoms (lasing) tab. There are several changes. The lamp has become a bank of lamps and they are now at right angles to the tube axis. The atom now has two excited levels. When you turn the lamp on, you will see some stimulated emission, but not a lot. If you decrease the lifetime of the upper level, the amount of stimulated emission increases and you start to see triplets of photons. Why does this happen? Consider what happens when a red photon goes near an atom. If the electron is in the ‘blue’ state, nothing happens. If the atom is in the ground state, there will be absorption. If the atom is in the ‘red’ state there will be stimulated emission. Decreasing the lifetime of the upper level causes there to be more electrons in the ‘red’ state than in the ground state. This is called a ‘population inversion’. This means, that when a red photon goes near an atom it is more likely that there will be stimulated emission than absorption.
So now, we have managed to get triplets and quadruplets of photons, but they are still travelling in random directions. This is why you need to add mirrors. Observe what happens when you ‘enable mirrors’. Any emission that happens to occur perpendicular to the mirrors, will bounce back and forth many times. Each pass allows the photons to cause more and more stimulated emission. Eventually the laser blows up! To fix this you just need to adjust the mirror reflectivity at one end. You can reduce it to zero for a really powerful pulsed laser or to some intermediate value for a continuous laser.
There you have it. To make a laser you need a lot stimulated emission in one direction, which you can get with a population inversion and a couple of mirrors.
Short Videos about Lasers
To complete your exploration of lasers you might want to watch two short videos.
Minute Physics: How lasers work (in theory) and
Smarter Everyday: How lasers work (in practice).
The Minute Physics video goes very fast — as usual — and introduces the statistical reason behind why stimulated emission happens and why the emitted photons are the same. The Smarter Every Day video says some irrelevant silly stuff but it also shows an open cavity laser that lets you start and stop the lasing with your hand.
I have attached a
student worksheet for a lesson on this topic for grade 12 physics and a second file that contains the
answers and extra information for teachers.
Make a Donation to PhET!
If you use PhET, be sure to make a donation. As well as providing financial support, your donation shows that teachers really value this tool and helps attract funding from corporations. Let’s all make sure that PhET it remains free for us and for the teachers and students around that world that lack our resources.
Tags: Energy, Light, Modern Physics, Nobel Prize, Quantum



