Chris Meyer, OAPT Past President, TDSB hybrid teacher-coach
Christopher.meyer@tdsb.on.ca
Back in the day, I used to teach at the Ontario Science Centre and present their school programs. I would meet the visiting teacher and mob of grade 9 students in advance of my “Cosmic Connections” program and ask, “So has your class covered the astronomy unit yet?” On more than one occasion, the teacher answered, “No. You’re it!” That’s right; my 45-minute extravaganza was all the astronomy that students would get in grade 9. Despite fifteen years passing since then, the topic of astronomy still does not fall within every teacher’s comfort zone, so I hope that sharing the resources for our inquiry-based unit on grade 9 astronomy will help. In this article I will explain the ideas and pedagogical design of our unit and hopefully encourage you to check it out!
This is the second part of Chris’s article on teaching astronomy by inquiry. For the first part, please go
here.
Now back to astronomy … or perhaps chemistry
I teach the astronomy unit right after our chemistry unit because there are so many chemistry ideas that astronomy uses and reinforces: atoms and spectroscopy; the particle theory of matter; the structure of the nucleus; and the origin of the elements of the periodic table. In fact, the story of our universe is the story of the periodic table, of the creation of the elements that our familiar world is made of. But before we can do chemistry in space, we need to understand how light encodes chemistry and that begins with heat!


Fun Question: Why do most people associate blue colours with cold temperatures?
Fun Question: Which part of the nail is warmer, the red part or the white part?
I introduced emission spectra for elements back in our chemistry unit, so this becomes a great opportunity to review that and built upon it as we introduce absorption spectra.
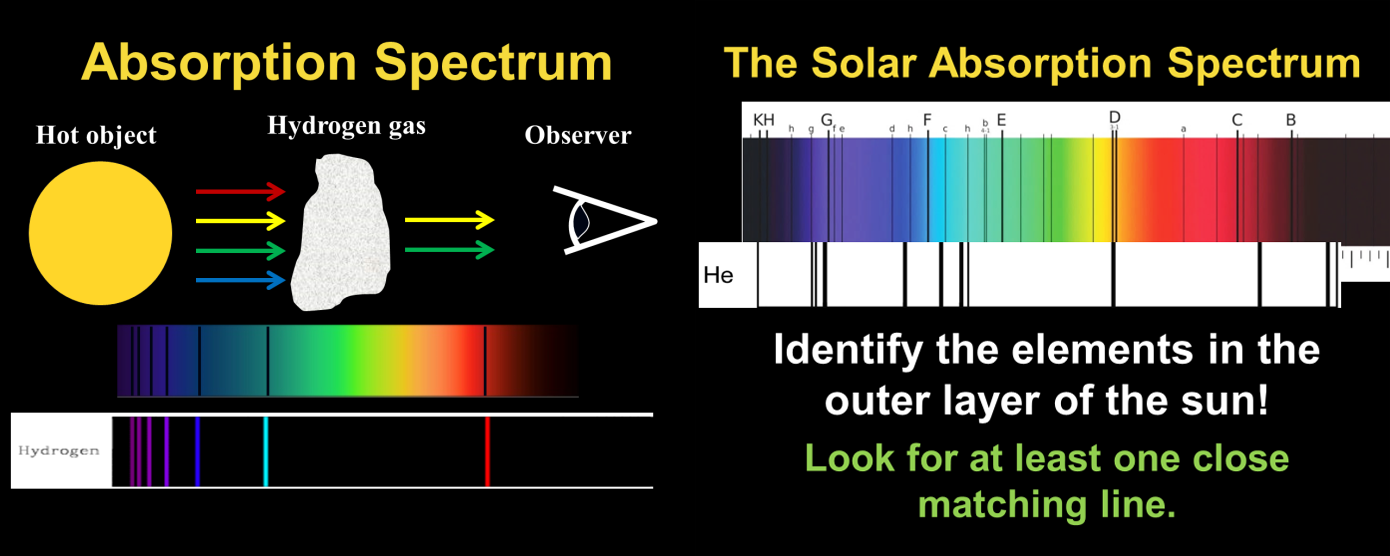

Most of our understanding of what stars, galaxies, and distant planets are composed of comes from the mysterious black lines that elements and molecules introduce into the spectrum of light. The study of the spectra of light, called spectroscopy, is right up there in the pantheon of “really important science” because it led to the foundation of quantum mechanics and our understanding of the stars! This is the first of many examples in astronomy where the science of the small (atoms) unravels the mysteries of the enormous (stars, galaxies, universe).
Science begins with evidence
Students have no good reason to believe anything we just tell them in a science class. Good science students should refuse to! If we are trying to do anything other than indoctrinate them into a cult, we must begin and end with evidence. Students need to understand how and why scientific knowledge was developed and go through that process themselves; they shouldn’t only be told the end results. New scientific ideas often begin with a surprising observation, followed by evidence gathering in search of a pattern and then new predictions tested by evidence. I might be guilty of a bit of hyperbole here, but where ever possible, we should present students with evidence for each new idea we share. Sometimes that evidence can come from things we observe and measure directly in the class. Other times it might need to be simulated evidence or reproductions of other people’s evidence. For example, we use a diffraction grating to take a careful look at the light from a warm bulb while changing its temperature. I have made videos of
these observations in case teachers don’t have access to the equipment.


Science’s explanatory power
Another concept at the heart of doing science is explaining new things using familiar, well-tested ideas. For those new to astronomy, everything can look unusual and different, but a closer examination will show that there are a lot of familiar science ideas at work. The very power of science comes from our ability to use a small number of simple ideas to explain a vast number of complex things. Newcomers to astronomy can be overwhelmed by the complexity of things in space. Our job as teachers and practitioners of science is to help students see the
ideas behind the complexity and draw connections between new phenomena and familiar knowledge. Otherwise, learning science is just memorizing lists of facts. The design of our astronomy unit is inspired by this obligation: trim out some of the complexity and focus on exploring the underlying ideas. This leads us to a great application of chemistry ideas that explains a lot about how a star works.
So, how does a star work?
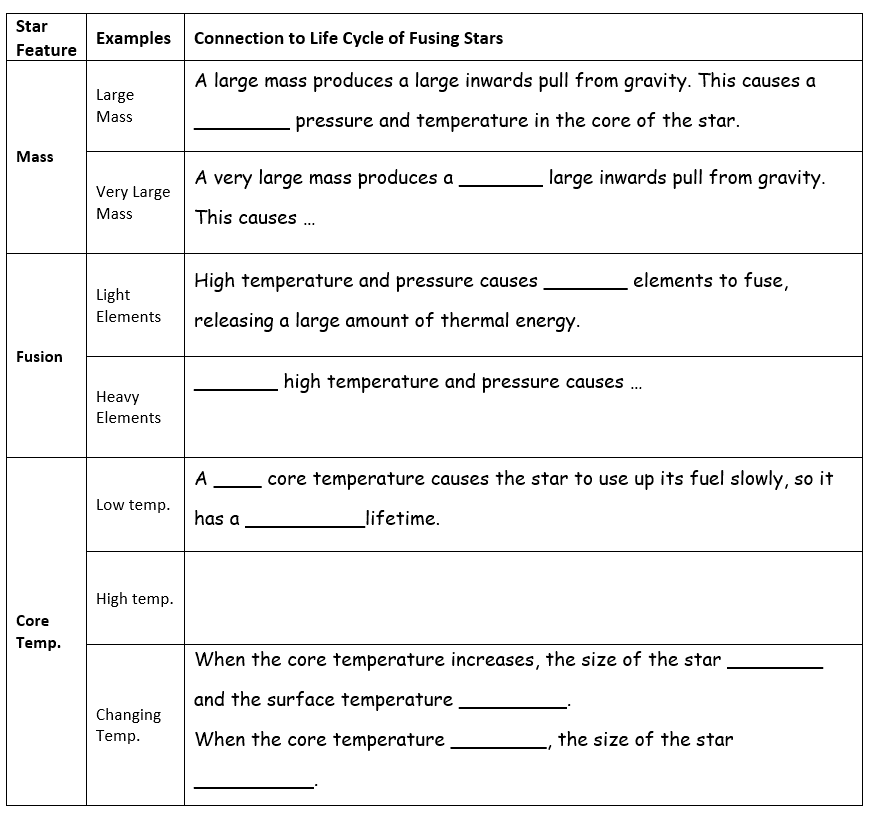
The insights from spectroscopy allow us to build a model of a star as a ball of very hot, ionized hydrogen gas. Each star experiences a tug-of-war between two factors that influence the course of its entire life: an inward pull due to the force of gravity and an outwards push from the thermal motions of the ions that compose it. This is a great opportunity to review the particle theory of matter and use it to explain the connection between the core temperature of a star and its size – an important part of explaining how a star changes over its lifetime.

 
Fun Question: Is a star alive?
The life of a star — is it alive?
No, it is not. But, we often talk about them as if they are! Stars are “alive” while they are able to replenish the thermal energy they lose to space through the process of nuclear fusion (usually, the joining of hydrogen nuclei). Here is a great opportunity to review atomic notation, as we explore the combination of protons and neutrons near the core of a star. This is also the first step in explaining the origin of the elements of the periodic table beyond helium. They come from the nuclear fires in the core of a star!
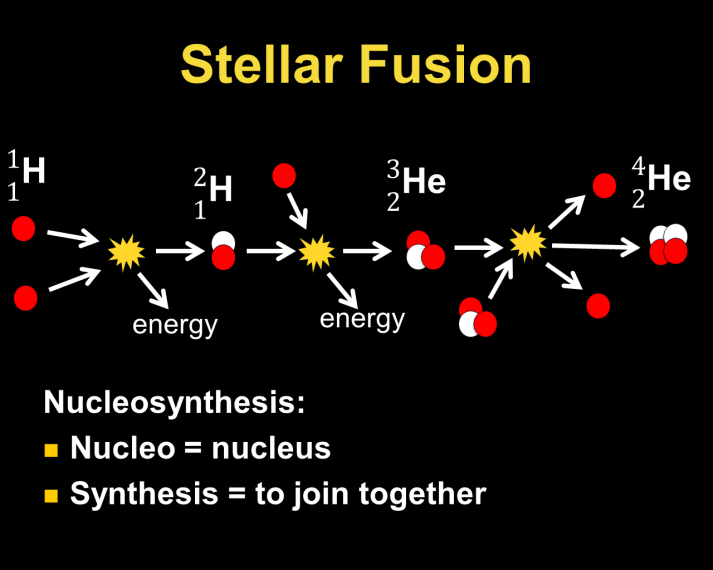
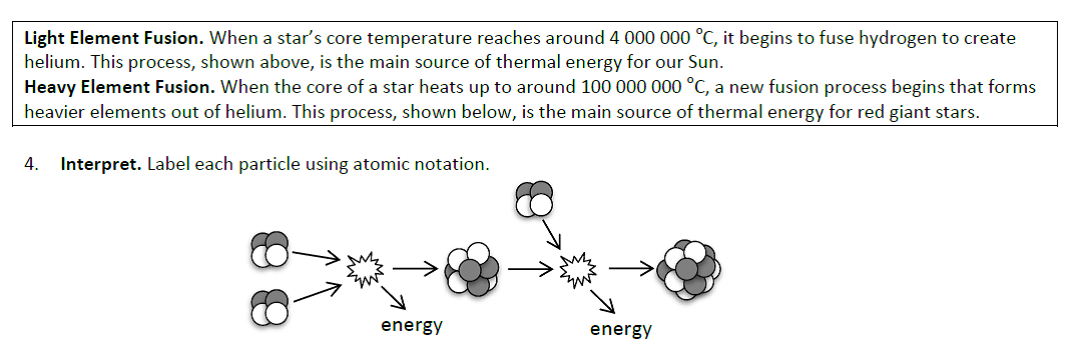
The phases in the life of a star are defined by the fusion processes at work. When a star runs low on hydrogen to fuse, it begins to fuse helium to other nuclei and manufacture elements in the periodic table up to iron. This process requires much greater core temperatures, causing the star to expand in size due to faster thermal motions of its particles (the particle theory, once again!). Unfortunately for the star’s aspirations, fusing iron nuclei will not release energy so the fusion process in the core stops and the star “dies”. We make use of
resources from the Perimeter Institute to explore these stages in the lifecycle of a star. Students enjoy the activity of piecing together the different evolutionary paths in the life of a star using their star cards.

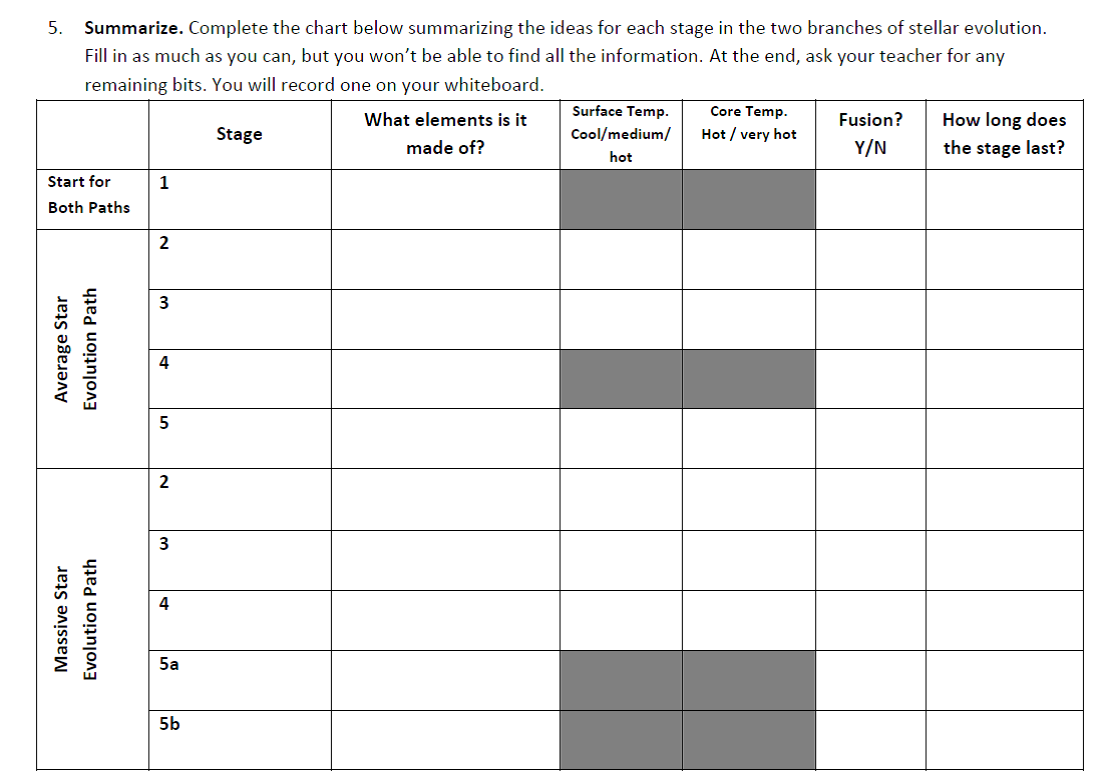
To prevent the activity from becoming an exercise in fact collecting, our lesson helps students focus on the underlying ideas: temperature, fusion, and the formation of new elements.

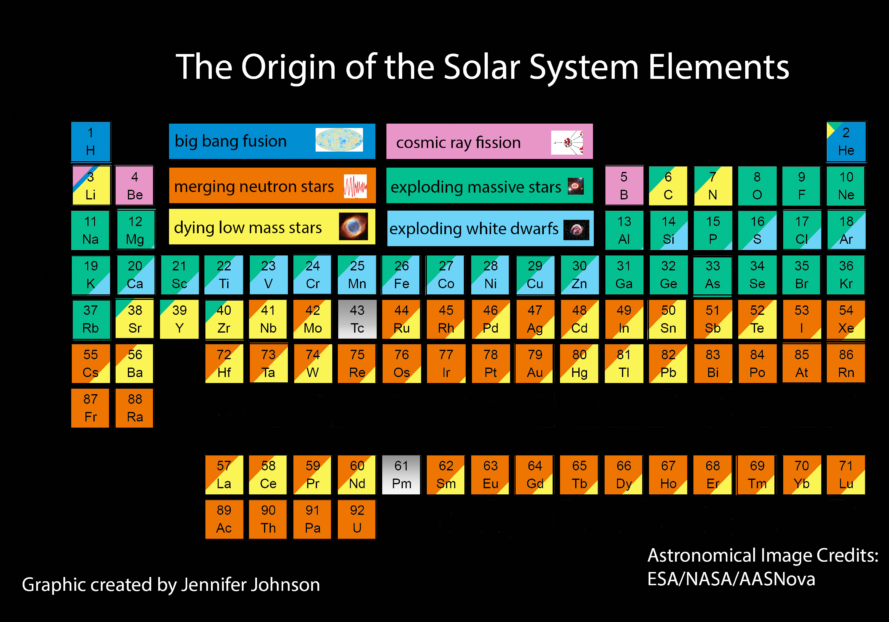
The Birds and the Bees of the Periodic Table
The full story of the periodic table is complex and fascinating. The Big Bang produced lots of hydrogen and helium, and not much else. Average and massive stars manufacture different elements by fusing nuclei in their core and other exotic processes. The violent death of massive stars in a supernova explosion and the spiralling coalescence of a pair of neutron stars produced more elements still! All of this provides an amazing perspective on us Earth-dwellers: the very matter we are made of was created or shared by the death of average stars, the supernova explosions of massive stars, and spiralling neutron stars, all of which must have happened in our solar neighbourhood before the formation of our Sun and solar system.

The Story of the Universe
The scientific story of the universe begins with a surprising observation: the absorption lines in light spectra from distant galaxies are out of place! They appear shifted compared with our Sun. This is a mystery to solve!


Fun Question: In what way have the absorption lines for the supercluster shifted?
The Perimeter Institute has put together a
good set of simulated spectrum data that helps students unravel this mystery. It turns out the shift in the lines is related to the speed and distance of the galaxies. We can visualize this result:


Results from spectra show galaxies moving away from us with the more distant ones moving fastest. If we take this snapshot and turn it into a movie, we could watch these galaxies moving away from us. Run that movie backwards and a shocking, new idea is suggested: earlier in time, all the matter and space we see in the universe came from one point. It took about 30 more years of hard work and evidence gathering before scientists became convinced of the Big Bang theory for the origin of our universe. I think it is remarkable that humans have put together a theory that successfully predicts the behaviour of the universe back in time by 13.8 billion years. In class, when we explore the history of the universe, we follow a similar strategy as we did with the lifecycle of stars: start with the Perimeter Institute’s handy
stages of the universe cards, collect the information, and then look for the important patterns that define the evolutionary life of the universe.


The forest for the trees, the stars for the galaxies
Maybe those should be song lyrics? Anyway, I cannot overemphasize the importance of exploring the ideas that underlie the complex phenomena we study in astronomy. Those ideas are what I want my students to learn, such as the ones neatly summarized in the Big Bang box in the example above. I don’t want them memorizing lists of information. This is why I feel so sad when I see most textbooks on this topic. I have one such example opened on my desk right now. In it I see reams of information about the life cycle of stars, all the phases of the moon labelled, diagrams labelling different layers in the star, classifications of different galaxy types, and lots of terminology that is used just once. This encyclopedic approach to teaching astronomy, or any topic, makes sense for the teacher — it is a careful presentation of all the relevant facts and ideas that person knows — but it doesn’t make sense considering how novices learn. If you are going to trouble students to learn something, make sure it appears again and again — no one-offs! Otherwise, students end up learning a set of isolated, disjoint facts that they can only memorize. But if we help them find the scientific threads that connect diverse phenomena, students begin to understand and that is the goal of science: to lay bare the universe to human understanding.
Final advice for teaching astronomy
When I design new lessons, I ask myself two questions: (1) how did people ever figure this stuff out; and (2) how do people actually use this knowledge? My hope is that you will find the answers to these questions within the lessons we use to teach astronomy. I encourage you to explore the teaching resources my school uses for the astronomy unit. They can be found on
my website. I have included the student investigation sheets, the PowerPoint course guide, and lots of videos that show the experiments and demonstrations in case you don’t have the equipment handy at your school. You can use these resources to build your personal understanding, explore how the content is organized, find new activities or demos, or use them as-is with your classes. If you have any questions, please feel free to
contact me. Good luck with your teaching!
Fun Answers
- Probably because ice looks blue-ish or people turn blue-ish when cold.
- The white part of the nail.
- The pattern of absorption lines has shifted to the right.
Tags: Astronomy, Light, Pedagogy














